Hypersensitivity reactions to intravenous iron: guidance for risk minimization and management | Haematologica

Safety and effectiveness of intravenous iron sucrose versus standard oral iron therapy in pregnant women with moderate-to-severe anaemia in India: a multicentre, open-label, phase 3, randomised, controlled trial - The Lancet Global

Management of Hypersensitivity Reactions to Nondextran Iron Products: New Insights Into Predisposing Risk Factors - ScienceDirect

Diagnostics | Free Full-Text | Compression of the Lateral Antebrachial Cutaneous Nerve due to Leakage of Iron after an Intravenous Iron Infusion
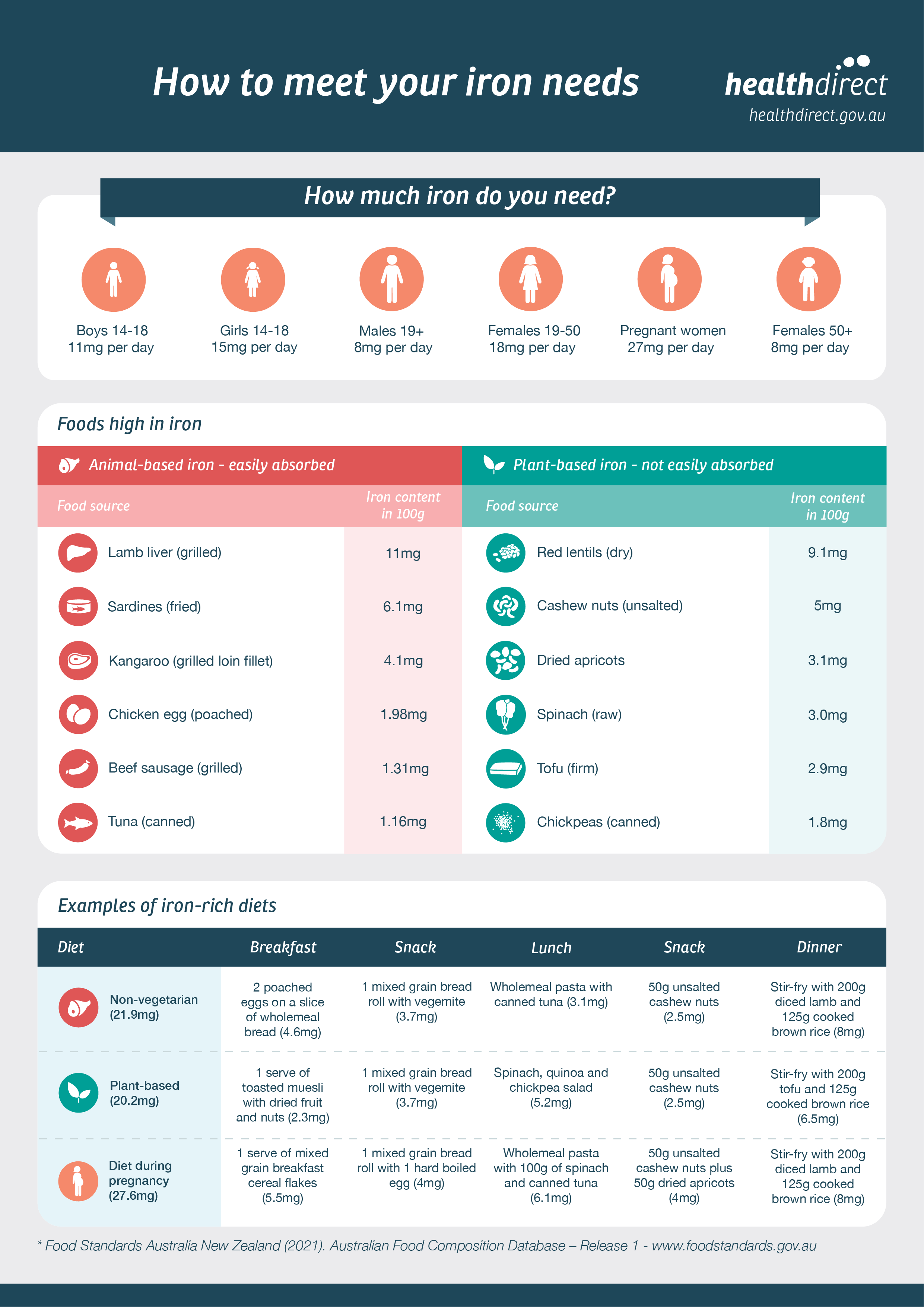
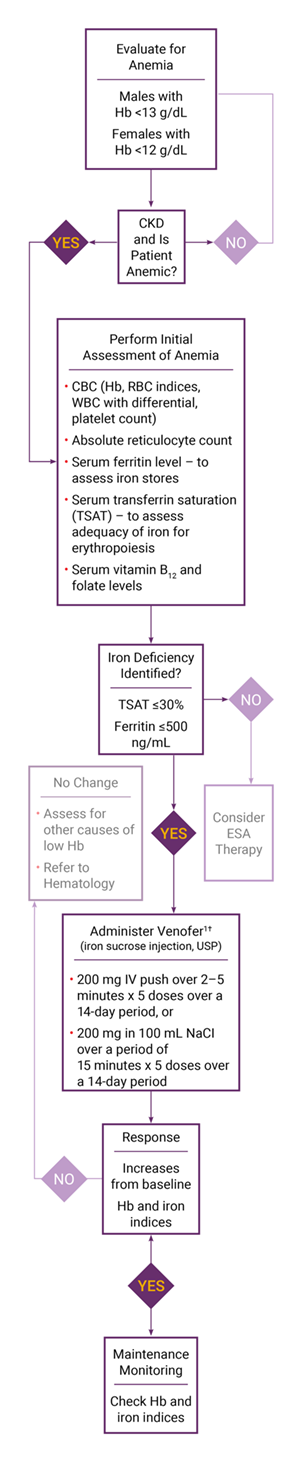
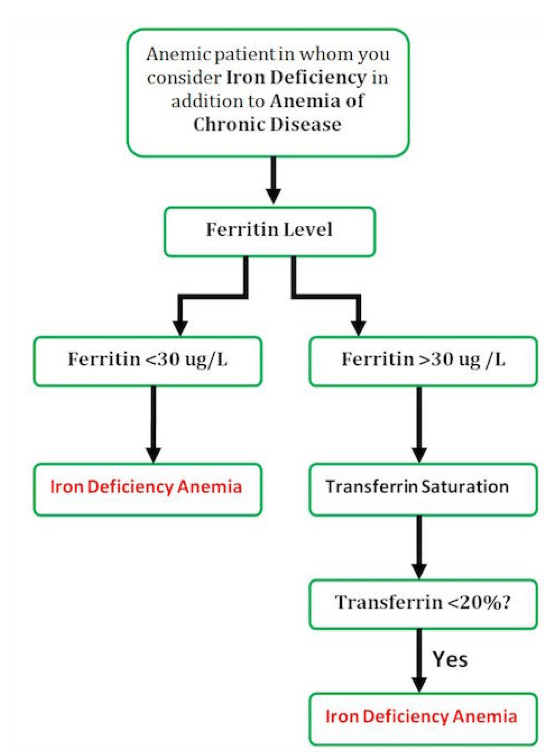
British Society of Gastroenterology guidelines for the management of iron deficiency anaemia in adults

Protocol for the cost-effectiveness of parenteral iron and oral iron therapy for first-line management of anaemia among pregnant women in a natural programme setting in Gujarat | BMJ Open
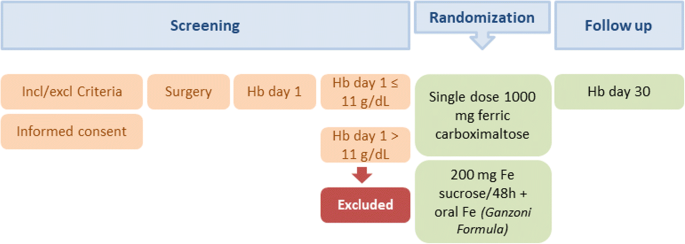
Single dose of intravenous ferric carboxymaltose infusion versus multiple fractionated doses of intravenous iron sucrose in the treatment of postoperative anaemia in colorectal cancer patients: study protocol for a randomised controlled trial

Effect of acute iron infusion on insulin secretion: A randomized, double-blind, placebo-controlled trial - eClinicalMedicine

Intravenous ferric derisomaltose in patients with heart failure and iron deficiency in the UK (IRONMAN): an investigator-initiated, prospective, randomised, open-label, blinded-endpoint trial - The Lancet